��ƽ�ĸ�������ҹ̶���øԭ���о�-985��ҵ������
��Դ��985������ ����ʱ�䣺2020-01-02 14:43
ժ Ҫ ���Ź̶���ø�������������죬���Ҽ����õ��˳���ķ�չ���봫ͳ��������������ͬ�����½�����һ��“��Ȼ��”���ҹ̶���ø�����¼���---��ƽ�ĸ�������ҹ̶���ø�����������˸ù̶�������ԭ����ʵ�����ü����ŵ����ڣ� �̶���ø����װ“��Ȼ”��ɣ�������Ȼ�����ԣ���С��һ�����ظ�ʹ�ã���ɫ��������ʵ�ֶ���ø�Ĺ�ͬ�̶��������Ӧ��ǰ��������չ����
����
�����ؼ��� �̶���ø�� ���Ҽ����� ��ƽ�ĸ���ӹ̶���ø�� “һ�߶�ø”����
����
�����̶���ø�����ѳ�Ϊø�����о����ص���ȵ�֮һ��������ø��ȣ��̶���ø�����ø���ȶ��ԣ���ʹø�ܹ������������á����Ҽ����ƶ��˹̶���ø�Ĺ㷺Ӧ�á����ҹ̶���ø���ø߷��Ӳ��Ͻ�ø�����ڽ�����Χ�γ�С�����������������ʳ�Ϊ���Ļ�о�ģ��ڿdz�Ϊ�ұڻ�ڲ�[1].���ҹ̶���ø�ܹ��ṩһ���ض��ķ�Ӧ������ʹ��Ӧ������ѡ���Խ�����ͳ�[2].���ҹ̶���ø������ɫ�������ܽ����ں�����ռ���룬�����ø���ȶ��ԣ����ҿɶ��������������ѧ���ʽ�����Ч���ء����ü����ѹ㷺Ӧ����ʳƷ��ҽҩ����ױƷ����������֯�����ũҵ������[3].Ȼ����ͳ�����ҹ̶���ø��������֮�����ڣ� ��ø����������Լ�����װ�豸�����������������ɱ��������ǶԲ���Ҫ������Ҫ�����豸���мӹ���ø���ң���ɱ����ߣ� ���ҿ�����һ��Ƿ�ѣ���Ʒ���ܲ���ϴ�[4]; �̶������̲����к�����[5],��������ȫ�������ȴ��������� �ȵȡ�����о�̽���µ�ø�̶��������������ȶ�����Ч������������������ʽ���������ҹ̶���ø��Ʒ���ܡ����������ɱ����������������е���Դ���ĺͻ�����Ⱦ�ȣ������ҹ̶���ø��������Ҫ�о����ݣ����ϵ���ҵ��ɫ�������ɳ�����չ��Ҫ��[6 -7].
����
����������ƽ�ĸ���ӵ��о�����֮ǰ���б��������ǽ�����Ӧ����ø�Ĺ̶���ȴ���б�������Щ���������Ŷ���ƽ�ĸ���ӵ������о�[8 -9],���ӵ�Ӧ�������õ��ؿ�����ƽ�ĸ�������ҹ̶���ø�� Saccharomyces cerevisiae spore microencapsulated en-zyme�� ����Ӧ�˶������ü��������û��̼������������Է��ڵ��ף� ��Ŀ��ø�ࣩ �����������н��б���ڲ��߹�����ʵ�ֶԸõ������ҹ̶������ü�������ƽ̨���跢���豸��������ģ�ɸ��ݲ�Ʒ�����������ڡ����ڶ���ƽ�ĸ���ӱڽṹ���о������о����Ѿ��ɹ�ʵ���˶���Ŀ��ø��Ľ�ĸ�������ҹ̶���[10 -12].����������ƽ�ĸ�������ҹ̶���ø�� ���¼�����ӹ̶���ø�� ��ԭ�������ӵ���װ�����Լ����ӹ̶���ø�����ƽ�����ϸ������������Ӧ��ǰ������չ����
����
������ĸ���ӹ̶���ø�ķ�����Ҫ���������֣� ����ѧ�����ͻ�ѧ�������Ǿ���[13]�ǹ̶���ø�ij������壬Ҳ��Ұ���ͽ�ĸ���ӱڵ���Ҫ��ɳɷ֣���ѧ������ͨ����������Ŀ��ø�̶���“�Ǿ�����”�ϣ���Ŀǰ���õĿǾ��ǹ̶���ø�����������ƣ� ��֮ͬ�����������������γɴ�С��һ����״�ȶ�����Ȼ��“�Ǿ�����”,Ȼ��Ŀ��øͨ�����������̶���“�Ǿ�����”�ϡ���ˣ����ĶԻ�ѧ�����ӹ̶���ø��ԭ��������ϸ������������˵��������ѧ������ͨ�����̼���������Ҫ����Ŀ����Է�����ø�������γɹ�����ֱ�ӹ̶������ӱ��У��γ���Ȼ�����ӹ̶���ø���ҡ��ü�������Ҫ���ʻ��������ӱ��еĿǾ��Dz�Ͷ��Ұ���㡣���£��������ӹ̶���ø����������ѧԭ����ʵ��������ϸ������
����
����1 ��ƽ�ĸ�������ҹ̶���øԭ����
����
������ƽ�ĸ������ϸ����Ӫ���ḻ��������ͨ��������˿���Ѳ����Ӵ����塣���ܵ�����в��ʱ�����絪Դȱ��������ʹ��ĸӪ��ϸ������˿���ѽ����������[14 -18],�������߳��������γ�һ������ 4��������ϸ�������ң�ÿ��������ϸ����Ϊ���ӡ��ڲ��߹����У��γ���ǰ����Ĥ��������Ĥ�ṹ�����ڵķ���ϵͳͨ���ع����������Է��ڵ���ת����ǰ����Ĥ�ڣ� ��ǰ����Ĥ���γ����ӱ�ʱ�������Է��ڵ��ױ����������ӱ��ڲ�����Ȼ��ǰ����Ĥ����Ĥ�����ӱڵ���װ��������ʧ�����ǣ���������ӱ���Ȼ�ܹ��������Է��ڵ������̶��ڱ��ڣ�����Ҫ�����ڿǾ��Dz�Ͷ��Ұ����Ĵ��ڡ�SUDA ��[19]�����ӱ�������Ұ������ƽ�ĸ�����ڱ����������ɫӫ�⵰�� GFP ʱ���֣����ܱ��̶������ӱ��ڣ� ����ȱ���˶��Ұ����� dit1Δ ����ȴ���ܡ���ˣ��������У����Ұ�����ܹ��赲�����͵�����ɢ��
����
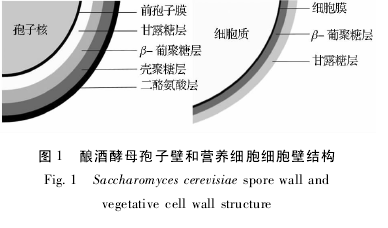
������ƽ�ĸ���ӱڰ�����������̬ѧ�ϲ�ͬ�Ľṹ��Σ����ڲൽ�������Ϊ��¶�Dz㣨 Mannan�� ��β-�Ͼ��Dz㣨 β-glucan�� ���Ǿ��Dz㣨 Chitosan�� �Ͷ��Ұ���㣨 Dityrosine��[9].�� 4 ��ṹ�ĺϳ����Դ��ڵ����˳�����ν��еġ����磬�������Ұ����ĺϳ�ֻ���ڴ����Ǿ��Dz�ϳ���Ϻ�ſ�ʼ�ϳ�[20].���ڿǾ��Dz�ȱʧ����ϳ�������ȱ�ݵľ��꣬���Ұ���㽫���ܺϳɡ�Դ�����ӱ����еĽṹ��Σ���ĸ���Ӿ��и߶ȵĿ����ԣ����翹����ø�����л��Լ������ȵ�[21 -22],��ˣ������ӽ������Է��ڵ��̶������ӱڵ�ͬʱҲ�������Ը����˹̶������ӱ��ڲ��ĵ��ס���ĸ���Ӳ��߹����п����Ե��ķ��ڱ�����Ƽ����ӱڵ�����ṹ��Σ������ǿǾ��Dz�Ͷ��Ұ����Ĵ�����ʵ�����ӹ̶���ø�����Ļ����� ����ڶ��Ұ���㣬�Ǿ��Dz����ø�Ĺ̶����ӱز����١�����ƽ�ĸӪ��ϸ��ϸ�������ɣ��ṹ�����ڶ��������� β-�Ͼ��Dz㣨 β-glu-can�� ��¶�Dz㣨 Mannan�� ��ɣ��Ǿ��Ǻ������٣����߱����Ұ���㣬��˶Կ����Է��ڵ��Ĺ̶�������Ҫ�ٺܶ�[10].��ƽ�ĸ���ӱں�Ӫ��ϸ��ϸ���ڽṹ��ͼ 1 ��ʾ��
����
������ƽ�ĸ�������ҹ̶���ø����ָ�� ͨ����������ѧ�ֶ��������б���Ŀ����Է��ڵ��ף��ڱ�ת���������ⲿʱ�������ӱڣ������ӱڵ�ijЩ����������� һ������Ƕ�ڿǾ��Dz��У�һ���ֱ��������Ͼ��Dz�Ͷ��Ұ����֮��Ŀռ��У� �������ӵij��죬�γ�����Ȼ����һ���ȶ������ӹ̶���ø��ͬʱ���������ӱ��������Ұ��������״����Ĥ������������絰���ʵȲ���ͨ������С�������������Ͳ���ȿ�������ͨ����������̶������ӱ��ڲ���ø����ϣ��Ӷ�ʵ�����ӹ̶���ø�Ե����ø�⡣
����
����
�����ؼ��� �̶���ø�� ���Ҽ����� ��ƽ�ĸ���ӹ̶���ø�� “һ�߶�ø”����
����
�����̶���ø�����ѳ�Ϊø�����о����ص���ȵ�֮һ��������ø��ȣ��̶���ø�����ø���ȶ��ԣ���ʹø�ܹ������������á����Ҽ����ƶ��˹̶���ø�Ĺ㷺Ӧ�á����ҹ̶���ø���ø߷��Ӳ��Ͻ�ø�����ڽ�����Χ�γ�С�����������������ʳ�Ϊ���Ļ�о�ģ��ڿdz�Ϊ�ұڻ�ڲ�[1].���ҹ̶���ø�ܹ��ṩһ���ض��ķ�Ӧ������ʹ��Ӧ������ѡ���Խ�����ͳ�[2].���ҹ̶���ø������ɫ�������ܽ����ں�����ռ���룬�����ø���ȶ��ԣ����ҿɶ��������������ѧ���ʽ�����Ч���ء����ü����ѹ㷺Ӧ����ʳƷ��ҽҩ����ױƷ����������֯�����ũҵ������[3].Ȼ����ͳ�����ҹ̶���ø��������֮�����ڣ� ��ø����������Լ�����װ�豸�����������������ɱ��������ǶԲ���Ҫ������Ҫ�����豸���мӹ���ø���ң���ɱ����ߣ� ���ҿ�����һ��Ƿ�ѣ���Ʒ���ܲ���ϴ�[4]; �̶������̲����к�����[5],��������ȫ�������ȴ��������� �ȵȡ�����о�̽���µ�ø�̶��������������ȶ�����Ч������������������ʽ���������ҹ̶���ø��Ʒ���ܡ����������ɱ����������������е���Դ���ĺͻ�����Ⱦ�ȣ������ҹ̶���ø��������Ҫ�о����ݣ����ϵ���ҵ��ɫ�������ɳ�����չ��Ҫ��[6 -7].
����
����������ƽ�ĸ���ӵ��о�����֮ǰ���б��������ǽ�����Ӧ����ø�Ĺ̶���ȴ���б�������Щ���������Ŷ���ƽ�ĸ���ӵ������о�[8 -9],���ӵ�Ӧ�������õ��ؿ�����ƽ�ĸ�������ҹ̶���ø�� Saccharomyces cerevisiae spore microencapsulated en-zyme�� ����Ӧ�˶������ü��������û��̼������������Է��ڵ��ף� ��Ŀ��ø�ࣩ �����������н��б���ڲ��߹�����ʵ�ֶԸõ������ҹ̶������ü�������ƽ̨���跢���豸��������ģ�ɸ��ݲ�Ʒ�����������ڡ����ڶ���ƽ�ĸ���ӱڽṹ���о������о����Ѿ��ɹ�ʵ���˶���Ŀ��ø��Ľ�ĸ�������ҹ̶���[10 -12].����������ƽ�ĸ�������ҹ̶���ø�� ���¼�����ӹ̶���ø�� ��ԭ�������ӵ���װ�����Լ����ӹ̶���ø�����ƽ�����ϸ������������Ӧ��ǰ������չ����
����
������ĸ���ӹ̶���ø�ķ�����Ҫ���������֣� ����ѧ�����ͻ�ѧ�������Ǿ���[13]�ǹ̶���ø�ij������壬Ҳ��Ұ���ͽ�ĸ���ӱڵ���Ҫ��ɳɷ֣���ѧ������ͨ����������Ŀ��ø�̶���“�Ǿ�����”�ϣ���Ŀǰ���õĿǾ��ǹ̶���ø�����������ƣ� ��֮ͬ�����������������γɴ�С��һ����״�ȶ�����Ȼ��“�Ǿ�����”,Ȼ��Ŀ��øͨ�����������̶���“�Ǿ�����”�ϡ���ˣ����ĶԻ�ѧ�����ӹ̶���ø��ԭ��������ϸ������������˵��������ѧ������ͨ�����̼���������Ҫ����Ŀ����Է�����ø�������γɹ�����ֱ�ӹ̶������ӱ��У��γ���Ȼ�����ӹ̶���ø���ҡ��ü�������Ҫ���ʻ��������ӱ��еĿǾ��Dz�Ͷ��Ұ���㡣���£��������ӹ̶���ø����������ѧԭ����ʵ��������ϸ������
����
����1 ��ƽ�ĸ�������ҹ̶���øԭ����
����
������ƽ�ĸ������ϸ����Ӫ���ḻ��������ͨ��������˿���Ѳ����Ӵ����塣���ܵ�����в��ʱ�����絪Դȱ��������ʹ��ĸӪ��ϸ������˿���ѽ����������[14 -18],�������߳��������γ�һ������ 4��������ϸ�������ң�ÿ��������ϸ����Ϊ���ӡ��ڲ��߹����У��γ���ǰ����Ĥ��������Ĥ�ṹ�����ڵķ���ϵͳͨ���ع����������Է��ڵ���ת����ǰ����Ĥ�ڣ� ��ǰ����Ĥ���γ����ӱ�ʱ�������Է��ڵ��ױ����������ӱ��ڲ�����Ȼ��ǰ����Ĥ����Ĥ�����ӱڵ���װ��������ʧ�����ǣ���������ӱ���Ȼ�ܹ��������Է��ڵ������̶��ڱ��ڣ�����Ҫ�����ڿǾ��Dz�Ͷ��Ұ����Ĵ��ڡ�SUDA ��[19]�����ӱ�������Ұ������ƽ�ĸ�����ڱ����������ɫӫ�⵰�� GFP ʱ���֣����ܱ��̶������ӱ��ڣ� ����ȱ���˶��Ұ����� dit1Δ ����ȴ���ܡ���ˣ��������У����Ұ�����ܹ��赲�����͵�����ɢ��
����
������ƽ�ĸ���ӱڰ�����������̬ѧ�ϲ�ͬ�Ľṹ��Σ����ڲൽ�������Ϊ��¶�Dz㣨 Mannan�� ��β-�Ͼ��Dz㣨 β-glucan�� ���Ǿ��Dz㣨 Chitosan�� �Ͷ��Ұ���㣨 Dityrosine��[9].�� 4 ��ṹ�ĺϳ����Դ��ڵ����˳�����ν��еġ����磬�������Ұ����ĺϳ�ֻ���ڴ����Ǿ��Dz�ϳ���Ϻ�ſ�ʼ�ϳ�[20].���ڿǾ��Dz�ȱʧ����ϳ�������ȱ�ݵľ��꣬���Ұ���㽫���ܺϳɡ�Դ�����ӱ����еĽṹ��Σ���ĸ���Ӿ��и߶ȵĿ����ԣ����翹����ø�����л��Լ������ȵ�[21 -22],��ˣ������ӽ������Է��ڵ��̶������ӱڵ�ͬʱҲ�������Ը����˹̶������ӱ��ڲ��ĵ��ס���ĸ���Ӳ��߹����п����Ե��ķ��ڱ�����Ƽ����ӱڵ�����ṹ��Σ������ǿǾ��Dz�Ͷ��Ұ����Ĵ�����ʵ�����ӹ̶���ø�����Ļ����� ����ڶ��Ұ���㣬�Ǿ��Dz����ø�Ĺ̶����ӱز����١�����ƽ�ĸӪ��ϸ��ϸ�������ɣ��ṹ�����ڶ��������� β-�Ͼ��Dz㣨 β-glu-can�� ��¶�Dz㣨 Mannan�� ��ɣ��Ǿ��Ǻ������٣����߱����Ұ���㣬��˶Կ����Է��ڵ��Ĺ̶�������Ҫ�ٺܶ�[10].��ƽ�ĸ���ӱں�Ӫ��ϸ��ϸ���ڽṹ��ͼ 1 ��ʾ��
����
������ƽ�ĸ�������ҹ̶���ø����ָ�� ͨ����������ѧ�ֶ��������б���Ŀ����Է��ڵ��ף��ڱ�ת���������ⲿʱ�������ӱڣ������ӱڵ�ijЩ����������� һ������Ƕ�ڿǾ��Dz��У�һ���ֱ��������Ͼ��Dz�Ͷ��Ұ����֮��Ŀռ��У� �������ӵij��죬�γ�����Ȼ����һ���ȶ������ӹ̶���ø��ͬʱ���������ӱ��������Ұ��������״����Ĥ������������絰���ʵȲ���ͨ������С�������������Ͳ���ȿ�������ͨ����������̶������ӱ��ڲ���ø����ϣ��Ӷ�ʵ�����ӹ̶���ø�Ե����ø�⡣
����
���� ����
����
 ����
����
2 ���ӵ���װ���̡�
����
���������ӵ��γɹ����У�������Ĥ�����ӱڶ�����ĸ��ϸ����ϸ�����Դ�ͷ�ϳɵķ�ʽ���е�[8]
����
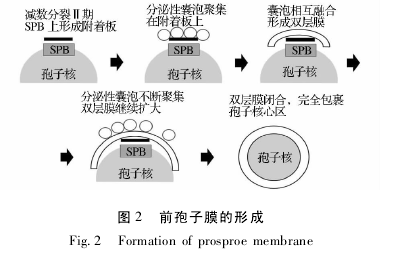
����.���ӵ��γ���Ҫ 2 ����Ҫ�IJ��裺 ��һ��ǰ����Ĥ���γɲ������������Ѣ��ڵ���ϸ�����γ�ǰ���ӣ� �ڶ������ӱڵ���װ��
����
����2. 1 ǰ����Ĥ��ǰ���ӵ��γɡ�
����
������ͼ 2 ��ʾ���ڼ������Ѣ��ڣ��ں˱�Ĥ������ �� �� �� �� 4 �� �� �� �� �� SPB �� spindle polebodies��[23 -24].���ݿ�ʼ�γɲ���λ�� SPB ����࣬�ڿ����� N-�һ��������ǰ����е������� SNARE�� soluble N-ethylmaleimide- sensitive protein recep-tor��[25]���������ںϣ��γɱ�ƽ��˫��Ĥ�ṹ������ṹ����ǰ����Ĥ�γɵĿ��ˡ����ż������Ѣ��ں˷��ѵĽ��У����ݲ��ϵ�����˫��Ĥ�ṹ���������������Ե���ϸ���ˡ��˷�����ɺ�˫��Ĥ���ս����Ե�ϸ���˺Ͳ���ĸϸ���е�ϸ���ʷ�����ڲ���������ԭ����ĸϸ���У������γɺ����ĸ�ǰ���ӵ����ҡ���ÿ�����ⲿ�γɵ�˫��Ĥ�ṹ��֮Ϊǰ����Ĥ[23 -24,26].
����
����
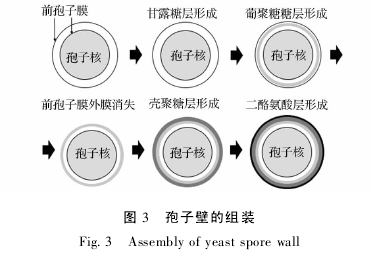
�������ӱڵ���װ����ǰ����Ĥ˫��Ĥ֮�����ǻ������ӱڲ��ϣ��ù��̵���ʼ������˫��Ĥ�ṹ����ǻ[27].��������ӱڵĹ����У�ǰ����Ĥ�����IJ���ϸ���ʽ��е����ʵĺϳɺͷ��ڣ������Է��ڵ�����ǰ����Ĥ���ڵ����ӱ���ࡣ�ڷ������Ƶ�Э�������£����ӱڴ��ڶ���������װ��ֻ�п��ڵ�һ�������װ�����һ��ſ�ʼ��װ�� ͼ 3�� : ���������ڲ��¶�Dz㣬���Ϊ β-�Ͼ��Dz㣬�ٴ�Ϊ�Ǿ��Dz㣬���Ϊ���Ұ����[20].һ����װ��ɣ����ӱڱ㸳������һϵ�п�������[28].
����
����
�������ӱںϳ���ʼ������Ļ���һ�㱻��Ϊ��GIP1 ����ͬʱ��ѧ�߷��� AMA1 ����Ҳ�����ӱںϳ���ʼ�������[29 -31].�� GIP1 ��ͻ�����ƣ�AMA1 ͻ��������ӱ����ϳ�[32 -33].�о�������AMA1 ���������ӱ��γɵ���ʼ�ͼ����ڶ��η�����ɵ��ν�����Э�����á�
����
����2. 2. 2 ��¶�Dz�� β-�Ͼ��Dz���γɡ�
����
����ǰ����Ĥ�պ�֮���������ݽ���¶�ǵ���������ǰ����Ĥ˫��Ĥ��ǻ�������������γɸ�¶�Dz㡣
����
����β-1,3-�Ͼ��Dz���γ���Ҫ β-�Ͼ��Ǻϳ�ø�IJ��룬��ø��������Ĥ�Ϻϳ� β-�Ͼ����������ͷ���ǰ����Ĥ����ǻ��Ȼ����Щ������һ��������װ�γ� β-�Ͼ��Dz㡣����ƽ�ĸ�У��� β-�Ͼ��Ǻϳ�ø�� 3 ���ǻ���ɣ����У�FKS1 ���������ǻ���Ҫ��Ӫ������ʱ�����ã�GSC2/FKS2 ���������ǻ���Ҫ�����ӱ���װ�������ã�Fks2 �Ǹ���ò�ϳɵ���Ҫ����[34].FKS3 ���������ǻ���������װ��һ������[35],�� fks3Δ ȱ�ݾ���չ�ֳ����ӱ�ȱ�ݵ���״[19].�����о��������������� β-�Ͼ��Dz���� �� �� װ �� �� �� �� Ҫ �� �ã��� SPO77,SPS2,SPS22,CRR1,SSP2[36 -39].����������������дز������г�Ա�����磬SPS2,SPS22 �ֱ��� ECM33,PST1��أ�CRR1 ������ˮ��ø���� CRH1 �� CRH2 ��أ�GAS ��Ӫ��ϸ���ڵ��ϳɻ��� PIR ���[40].��Щ�������е�������װ��¶�Dz�� β-�Ͼ��Dz�ʱ���ӵ����õ�ͬ����Ӧ������Ӫ��ϸ���ڹ���ʱ���ӵ����á��� β-�Ͼ��Dz���װ��Ϻ�ǰ����Ĥ��Ĥ��ʧ�� ��Ȼ���������в������ ���Ǹ�Ĥ����ʧ����ʹijЩ���ӱ���װ������ Osw1 ���룬�Ӷ��γ��������[36,39]; ͬʱ����װ��ø�¶�Dz�� β-�Ͼ��Dz��ɲ�������⣬ʹ�������ڹ�ѧ�����¿ɼ���
���о���ĸ���ӹ̶���ø��ͬʱ�����Ƿ��֣� ���ڿǾ��Ǿ��������Ե��ص㣬��Ϊ�Ǿ������ dit1Δ���ӿ��������������Ӻ����Ի�������磬���о����ź���[11]�����Ұ����ȱʧ�� dit1Δ ���������Ǿ������������� Cu2 +,�������ӱ�������Ұ���ͺ�ͬʱȱʧ���Ұ����ͿǾ��Dz�� chs3Δ ���ӽ��бȽϡ������ʾ����Ϊ�Ǿ������ dit1Δ ���ӣ����� Cu2 +������ǿ�� ��������ӱ�������Ұ���ͣ� ��ȱʧ�˶��Ұ����ͿǾ��Dz�� chs3Δ �������� Cu2 +����������֮���ֶ��������� Cr3 +��Ni2 +��Zn2 +��Cd2 +��Pb2 +�����ӵ������������о���ͬ������Ϊ�Ǿ������ dit1Δ ���ӣ��Ը������ӵ�������������ϸ����������ߵģ���Ӫ��ϸ������͵ģ� ������ȱʧ������ 2 ��� chs3Δ ���ӣ���������Ҳ��Ӫ��ϸ����2 ���ࡣ�ɴ�˵����ȱʧ�˶��Ұ����� dit1Δ ������Ϊ�Ǿ����������ؽ������ӵ����������ѡ��ͬʱ�����ڿǾ��Ǵ����磬�ֶ���Ϊ�Ǿ������ dit1Δ�����������и���ɵ�ţ�ǵ������������о����������������ӽ�����ƣ�dit1Δ ��������ţ�Ƶ�����������Ұ�������Ӻ� chs3Δ ���ӡ���������ʵ��˵�� dit1Δ ������Ϊ�Ǿ�������ؽ�����ijЩ����ɷ��ӵ������ǿ��еġ����о�����ƽ�ĸ���ӵ�Ӧ�ôӹ̶���ø�����һ����չ���˻�������Ϊ��ƽ�ĸ���ӵ�Ӧ�ÿ����˵�·��
����
�����������������˶����������������о��⣬��ʵ���Ҷ��ڽ�ĸ���ӹ̶���ø���о��ǽ�ĸ����Ӧ���о����ص㡣�������ҹ̶���ø�����ķ�չ����ƽ�ĸ���ӹ̶���ø��Ϊ���ҹ̶���ø�������³�Ա��������ص��ŵ��״�չ���ڴ����ǰ�� �̶���ø����װ“��Ȼ”��ɣ�������Ȼ�����ԣ���С��һ�����ظ�ʹ�ã���ɫ������“һ�߶�ø��ϵ”�Ѿ������������ȵȡ����������ŵ㼰�о���������ţ���ĸ���ӹ̶���ø����Ϊø�����ҹ̶����������ʣ�
����
������ �� �� �ף�
����
����[1] BENITA S. Microencapsulation: methods and industrialapplications [M]. CRC Press,2005.
����[2] PEYRATOUT C S,DAEHNE L. Tailor-made polyelectro-lyte microcapsules: from multilayers to smart containers[J]. Angewandte Chemie International Edition,2004,43�� 29�� : 3 762 -3 783.
����[3] MEIER W. Polymer nanocapsules [J]. Chemical SocietyReviews,2000,29�� 5�� : 295 - 303.
����[4] Ҧ������÷�ֺ͡� һ����������������ϵ�����������ʹ̶��������е�Ӧ�� [J]. Ĥ��ѧ�뼼����1999,19�� 1�� : 19 - 23.
����[5] HANEFELD U,GARDOSSI L,MAGNER E. Understand-ing enzyme immobilisation [J]. Chemical Society Re-views,2009,38�� 2�� : 453 - 468.
����[6] ��ʫһ�� ��Դ���ģ�������̼�ŷ����й���ҵ�Ŀɳ�����չ[J]. �����о���2009�� 4�� : 41 -55.
����[7] ���ʣ������������������ȡ� ��̼���õķ�չģʽ�о�[J]. �й��˿� ��Դ�뻷����2008,18�� 3�� : 14 -19.
����[8] NEIMAN A M. Ascospore formation in the yeast Saccharo-myces cerevisiae [J]. Microbiology and Molecular BiologyReviews,2005,69�� 4�� : 565 - 584.
����[9] NEIMAN A M. Sporulation in the budding yeast Saccharo-myces cerevisiae [J]. Genetics,2011,189 �� 3 �� : 737 -765.
����[10] SHI L,LI Z,TACHIKAWA H,et al. Use of yeast sporesfor microencapsulation of enzymes [J]. Applied and en-vironmental microbiology,2014,80 �� 15 �� : 4 502 -4 510.
����[11] ZHANG H,TACHIKAWA H,GAO X-D,et al. Appliedusage of yeast spores as chitosan beads [J]. Applied andenvironmental microbiology,2014,80 �� 16 �� : 5 098 -5 105.
����[12] LI Z,LI Y,DUAN S,et al. Bioconversion of d-glucoseto d-psicose with immobilized d-xylose isomerase andd-psicose 3-epimerase on Saccharomyces cerevisiae spores[J]. Journal of industrial microbiology & biotechnology,2015,42�� 8�� : 1 117 - 1 128.
����[13] CHEN S-C,DUAN K-J. Production of Galactooligosac-charides Using β-Galactosidase Immobilized on Chitosan-Coated Magnetic Nanoparticles with Tris �� hydroxymethyl��phosphine as an Optional Coupling Agent [J]. Interna-tional journal of molecular sciences,2015,16 �� 6 �� :12 499 - 12 512.
����[14] HONIGBERG S M. Ime2p and Cdc28p: Co-pilots drivingmeiotic development [J]. Journal of cellular biochemis-try,2004,92�� 5�� : 1 025 - 1 033.
����[15] LEE B,AMON A. Meiosis: how to create a specializedcell cycle [J]. Current opinion in cell biology,2001,13�� 6�� : 770 -777.
����[16] MITCHELL A P. Control of meiotic gene expression inSaccharomyces cerevisiae [J]. Microbiological reviews,1994,58�� 1�� : 56 - 70.
����[17] ROEDER G S. Sex and the single cell: meiosis in yeast[J]. Proceedings of the National Academy of Sciences,1995,92�� 23�� : 10 450 - 10 456.
����[18] STERN B M. FEARless in meiosis [J]. Molecular cell,2003,11�� 5�� : 1 123 - 1 125.
����[19] SUDA Y,RODRIGUEZ R K,COLUCCIO A E,et al. Ascreen for spore wall permeability mutants identifies a se-creted protease required for proper spore wall assembly[J]. PLoS One,2009,4�� 9�� : e7184.
����[20] TACHIKAWA H,BLOECHER A,TATCHELL K,et al.A Gip1p-Glc7p phosphatase complex regulates septin or-ganization and spore wall formation [J]. The Journal ofcell biology,2001,155�� 5�� : 797 - 808.
����[21] EGEL R. Selective spore survival during replica-plating offission yeast [J]. Archives of microbiology,1977,112�� 1�� : 109 -110.
����[22] KUPIEC M,BYERS B,ESPOSITO R E,et al. 11 Meio-sis and Sporulation in Saccharomyces cerevisiae [J]. ColdSpring Harbor Monograph Archive,1997,21: 889 -1 036.
����[23] BYERS B. Cytology of the yeast life cycle [J]. ColdSpring Harbor Monograph Archive,1981,11: 59 - 96.
����[24] STRATHERN J N,JONES E W,BROACH J R. Molecu-lar biology of the yeast saccharomyces,life cycle and in-heritance [M]. Cold Spring Harbor Laboratory,1981.
����[25] NAKANISHI H,MORISHITA M,SCHWARTZ C L,etal. Phospholipase D and the SNARE Sso1p are necessaryfor vesicle fusion during sporulation in yeast [J]. J CellSci,2006,119�� 7�� : 1 406 - 1 415.
����[26] MOENS P B. Fine structure of ascospore development inthe yeast Saccharomyces cerevisiae [J]. Canadian Journalof Microbiology,1971,17�� 4�� : 507 - 510.
����[27] LYNN R R,MAGEE P T. Development of the spore wallduring ascospore formation in Saccharomyces cerevisiae[J]. The Journal of cell biology,1970,44 �� 3�� : 688- 692.
�� [28] SMITS G J,VAN DEN ENDE H,KLIS F M. Differentialregulation of cell wall biogenesis during growth and devel-opment in yeast [J]. Microbiology,2001,147�� 4�� : 781- 794.
����[29] COOPER K F,MALLORY M J,EGELAND D B,et al.Ama1p is a meiosis-specific regulator of the anaphase pro-moting complex / cyclosome in yeast [J]. Proceedings ofthe National Academy of Sciences, 2000, 97 �� 26 �� :14 548 - 14 553.
����[30] OELSCHLAEGEL T,SCHWICKART M,MATOS J,etal. The yeast APC / C subunit Mnd2 prevents prematuresister chromatid separation triggered by the meiosis-specif-ic APC / C-Ama1 [J]. Cell,2005,120�� 6�� : 773 - 788.
����[31] PENKNER A M,PRINZ S,FERSCHA S,et al. Mnd2,an essential antagonist of the anaphase-promoting complexduring meiotic prophase [J]. Cell,2005,120�� 6�� : 789- 801.
����[32] COLUCCIO A,NEIMAN A M. Interspore bridges: a newfeature of the Saccharomyces cerevisiae spore wall [J].Microbiology,2004,150�� 10�� : 3 189 - 3 196.meiotic exit to cytokinesis during sporulation in Saccharo-myces cerevisiae [J]. Molecular biology of the cell,2009,20�� 1�� : 134 - 145.
����[33] DIAMOND A E,PARK J-S,INOUE I,et al. The ana-phase promoting complex targeting subunit Ama1 links
����[34] HIMURO Y,TAKAI M,ISHIHARA K. Poly �� vinylferro-cene-co-2-hydroxyethyl methacrylate�� mediator as immo-bilized enzyme membrane for the fabrication of ampero-metric glucose sensor [J]. Sensors and Actuators B:Chemical,2009,136�� 1�� : 122 - 127.
����[35] DEUTSCHBAUER A M,WILLIAMS R M,CHU A M,etal. Parallel phenotypic analysis of sporulation and post-germination growth in Saccharomyces cerevisiae [J]. Pro-ceedings of the National Academy of Sciences,2002,99�� 24�� : 15 530 -15 535.
����[36] COLUCCIO A,BOGENGRUBER E,CONRAD M N,etal. Morphogenetic pathway of spore wall assembly in Sac-charomyces cerevisiae [J]. Eukaryotic Cell,2004,3�� 6�� : 1 464 -1 475.
����[37] GOMEZ-ESQUER F,RODR�PGUEZ-PE�rA J M,D�PAZG,et al. CRR1,a gene encoding a putative transglycosi-dase,is required for proper spore wall assembly in Sac-charomyces cerevisiae [J]. Microbiology, 2004, 150�� 10�� : 3 269 -3 280.
����[38] SARKAR P,FLORCZYK M,MCDONOUGH K,et al.SSP2, a sporulation-specific gene necessary for outerspore wall assembly in the yeast Saccharomyces cerevisiae[J]. Molecular Genetics and Genomics,2002,267�� 3�� :348 - 358.
����[39] LI J,AGARWAL S,ROEDER G S. SSP2 and OSW1,two sporulation-specific genes involved in spore morpho-genesis in Saccharomyces cerevisiae [J]. Genetics,2007,175�� 1�� : 143 - 154.
����[40] CHU S,DERISI J,EISEN M,et al. The transcriptionalprogram of sporulation in budding yeast [J]. Science,1998,282�� 5389�� : 699 - 705.
����[41] CHRISTODOULIDOU A,BOURIOTIS V,THIREOS G.Two sporulation-specific chitin deacetylase- encodinggenes are required for the ascospore wall rigidity of Sac-charomyces cerevisiae [J]. Journal of Biological Chemis-try,1996,271�� 49�� : 31 420 - 31 425.
����[42] MISHRA C,SEMINO C E,MCCREATH K J,et al. Clo-ning and expression of two chitin deacetylase genes ofSaccharomyces cerevisiae [J]. Yeast,1997,13�� 4�� : 327- 336.[43] PAMMER M,BRIZA P,ELLINGER A,et al. DIT101�� CSD2,CAL1�� ,a cell cycle �\ regulated yeast gene re-quired for synthesis of chitin in cell walls and chitosan inspore walls [J]. Yeast,1992,8�� 12�� : 1 089 - 1099.
����[44] SANZ M,TRILLA O A,DURAN N,et al. Control ofchitin synthesis through Shc1p,a functional homologue ofChs4p specifically induced during sporulation [J]. Mo-lecular microbiology,2002,43�� 5�� : 1 183 - 1 195.
����[45] NEUWALD A F. Barth syndrome may be due to an acyl-transferase deficiency [J]. Current Biology, 1997,7�� 8�� : R462 - R6.
����[46] BRIZA P,ELLINGER A,WINKLER G,et al. Charac-terization of a DL-dityrosine-containing macromoleculefrom yeast ascospore walls [J]. Journal of BiologicalChemistry,1990,265�� 25�� : 15 118 - 15 123.
����[47] BRIZA P,KALCHHAUSER H,PITTENAUER E,et al.N,N' Bisformyl Dityrosine is an in vivo Precursor of theYeast Ascospore Wall [J]. European journal of biochem-istry,1996,239�� 1�� : 124 - 131.
����[48] BRIZA P,ECKERSTORFER M,BREITENBACH M.The sporulation-specific enzymes encoded by the DIT1and DIT2 genes catalyze a two-step reaction leading to asoluble LL-dityrosine-containing precursor of the yeastspore wall [J]. Proceedings of the National Academy ofSciences,1994,91�� 10�� : 4 524 - 4 528.
����[49] FELDER T,BOGENGRUBER E,TENREIRO S,et al.Dtr1p,a multidrug resistance transporter of the major fa-cilitator superfamily,plays an essential role in spore wallmaturation in Saccharomyces cerevisiae [J]. Eukaryoticcell,2002,1�� 5�� : 799 - 810.
����[50] ���㡣 ��ƽ�ĸ���ӹ̶���ø�� D-�����Ǻϳ� D-����ͪ�ǵ��о� [J]. 2015.
����[51] CHOW C K,PALECEK S P. Enzyme encapsulation inpermeabilized Saccharomyces cerevisiae cells [J]. Bio-technology progress,2004,20�� 2�� : 449 - 456.
����[52] ROMERO C,SANCHEZ S,MANJON S,et al. Optimi-zation of the pectinesterase / endo-D-polygalacturonase co-immobilization process [J]. Enzyme and microbial tech-nology,1989,11�� 12�� : 837 - 843.
����[53] MEN Y,ZHU Y,ZENG Y,et al. Co-expression of d-glucose isomerase and d-psicose 3-epimerase: develop-ment of an efficient one-step production of d-psicose [J].Enzyme and microbial technology,2014,64: 1 - 5.
����[54] MASARU S,MITSUTAKA Y. A new enzymatic serumcreatinine measurement based on an endogenous creatine-eliminating system [J]. Clinica chimica acta,1984,143�� 2�� : 147 -155.
����[55] FOSSATI P,PRENCIPE L,BERTI G. Enzymic creati-nine assay: a new colorimetric method based on hydrogenperoxide measurement [J]. Clinical chemistry,1983,29�� 8�� : 1 494 -1 496.
����[56] COLUCCIO A E,RODRIGUEZ R K,KERNAN M J,etal. The yeast spore wall enables spores to survive passagethrough the digestive tract of Drosophila [J]. PLoS One,2008,3�� 8�� : e2873.
����
���������ӵ��γɹ����У�������Ĥ�����ӱڶ�����ĸ��ϸ����ϸ�����Դ�ͷ�ϳɵķ�ʽ���е�[8]
����
����.���ӵ��γ���Ҫ 2 ����Ҫ�IJ��裺 ��һ��ǰ����Ĥ���γɲ������������Ѣ��ڵ���ϸ�����γ�ǰ���ӣ� �ڶ������ӱڵ���װ��
����
����2. 1 ǰ����Ĥ��ǰ���ӵ��γɡ�
����
������ͼ 2 ��ʾ���ڼ������Ѣ��ڣ��ں˱�Ĥ������ �� �� �� �� 4 �� �� �� �� �� SPB �� spindle polebodies��[23 -24].���ݿ�ʼ�γɲ���λ�� SPB ����࣬�ڿ����� N-�һ��������ǰ����е������� SNARE�� soluble N-ethylmaleimide- sensitive protein recep-tor��[25]���������ںϣ��γɱ�ƽ��˫��Ĥ�ṹ������ṹ����ǰ����Ĥ�γɵĿ��ˡ����ż������Ѣ��ں˷��ѵĽ��У����ݲ��ϵ�����˫��Ĥ�ṹ���������������Ե���ϸ���ˡ��˷�����ɺ�˫��Ĥ���ս����Ե�ϸ���˺Ͳ���ĸϸ���е�ϸ���ʷ�����ڲ���������ԭ����ĸϸ���У������γɺ����ĸ�ǰ���ӵ����ҡ���ÿ�����ⲿ�γɵ�˫��Ĥ�ṹ��֮Ϊǰ����Ĥ[23 -24,26].
����
���� ����
����
����2. 2 ���ӱڵ���װ�� ����
��������
�������ӱڵ���װ����ǰ����Ĥ˫��Ĥ֮�����ǻ������ӱڲ��ϣ��ù��̵���ʼ������˫��Ĥ�ṹ����ǻ[27].��������ӱڵĹ����У�ǰ����Ĥ�����IJ���ϸ���ʽ��е����ʵĺϳɺͷ��ڣ������Է��ڵ�����ǰ����Ĥ���ڵ����ӱ���ࡣ�ڷ������Ƶ�Э�������£����ӱڴ��ڶ���������װ��ֻ�п��ڵ�һ�������װ�����һ��ſ�ʼ��װ�� ͼ 3�� : ���������ڲ��¶�Dz㣬���Ϊ β-�Ͼ��Dz㣬�ٴ�Ϊ�Ǿ��Dz㣬���Ϊ���Ұ����[20].һ����װ��ɣ����ӱڱ㸳������һϵ�п�������[28].
����
���� ����
����
����2. 2. 1 ���ӱںϳɵ���ʼ�� ����
��������
�������ӱںϳ���ʼ������Ļ���һ�㱻��Ϊ��GIP1 ����ͬʱ��ѧ�߷��� AMA1 ����Ҳ�����ӱںϳ���ʼ�������[29 -31].�� GIP1 ��ͻ�����ƣ�AMA1 ͻ��������ӱ����ϳ�[32 -33].�о�������AMA1 ���������ӱ��γɵ���ʼ�ͼ����ڶ��η�����ɵ��ν�����Э�����á�
����
����2. 2. 2 ��¶�Dz�� β-�Ͼ��Dz���γɡ�
����
����ǰ����Ĥ�պ�֮���������ݽ���¶�ǵ���������ǰ����Ĥ˫��Ĥ��ǻ�������������γɸ�¶�Dz㡣
����
����β-1,3-�Ͼ��Dz���γ���Ҫ β-�Ͼ��Ǻϳ�ø�IJ��룬��ø��������Ĥ�Ϻϳ� β-�Ͼ����������ͷ���ǰ����Ĥ����ǻ��Ȼ����Щ������һ��������װ�γ� β-�Ͼ��Dz㡣����ƽ�ĸ�У��� β-�Ͼ��Ǻϳ�ø�� 3 ���ǻ���ɣ����У�FKS1 ���������ǻ���Ҫ��Ӫ������ʱ�����ã�GSC2/FKS2 ���������ǻ���Ҫ�����ӱ���װ�������ã�Fks2 �Ǹ���ò�ϳɵ���Ҫ����[34].FKS3 ���������ǻ���������װ��һ������[35],�� fks3Δ ȱ�ݾ���չ�ֳ����ӱ�ȱ�ݵ���״[19].�����о��������������� β-�Ͼ��Dz���� �� �� װ �� �� �� �� Ҫ �� �ã��� SPO77,SPS2,SPS22,CRR1,SSP2[36 -39].����������������дز������г�Ա�����磬SPS2,SPS22 �ֱ��� ECM33,PST1��أ�CRR1 ������ˮ��ø���� CRH1 �� CRH2 ��أ�GAS ��Ӫ��ϸ���ڵ��ϳɻ��� PIR ���[40].��Щ�������е�������װ��¶�Dz�� β-�Ͼ��Dz�ʱ���ӵ����õ�ͬ����Ӧ������Ӫ��ϸ���ڹ���ʱ���ӵ����á��� β-�Ͼ��Dz���װ��Ϻ�ǰ����Ĥ��Ĥ��ʧ�� ��Ȼ���������в������ ���Ǹ�Ĥ����ʧ����ʹijЩ���ӱ���װ������ Osw1 ���룬�Ӷ��γ��������[36,39]; ͬʱ����װ��ø�¶�Dz�� β-�Ͼ��Dz��ɲ�������⣬ʹ�������ڹ�ѧ�����¿ɼ���
���о���ĸ���ӹ̶���ø��ͬʱ�����Ƿ��֣� ���ڿǾ��Ǿ��������Ե��ص㣬��Ϊ�Ǿ������ dit1Δ���ӿ��������������Ӻ����Ի�������磬���о����ź���[11]�����Ұ����ȱʧ�� dit1Δ ���������Ǿ������������� Cu2 +,�������ӱ�������Ұ���ͺ�ͬʱȱʧ���Ұ����ͿǾ��Dz�� chs3Δ ���ӽ��бȽϡ������ʾ����Ϊ�Ǿ������ dit1Δ ���ӣ����� Cu2 +������ǿ�� ��������ӱ�������Ұ���ͣ� ��ȱʧ�˶��Ұ����ͿǾ��Dz�� chs3Δ �������� Cu2 +����������֮���ֶ��������� Cr3 +��Ni2 +��Zn2 +��Cd2 +��Pb2 +�����ӵ������������о���ͬ������Ϊ�Ǿ������ dit1Δ ���ӣ��Ը������ӵ�������������ϸ����������ߵģ���Ӫ��ϸ������͵ģ� ������ȱʧ������ 2 ��� chs3Δ ���ӣ���������Ҳ��Ӫ��ϸ����2 ���ࡣ�ɴ�˵����ȱʧ�˶��Ұ����� dit1Δ ������Ϊ�Ǿ����������ؽ������ӵ����������ѡ��ͬʱ�����ڿǾ��Ǵ����磬�ֶ���Ϊ�Ǿ������ dit1Δ�����������и���ɵ�ţ�ǵ������������о����������������ӽ�����ƣ�dit1Δ ��������ţ�Ƶ�����������Ұ�������Ӻ� chs3Δ ���ӡ���������ʵ��˵�� dit1Δ ������Ϊ�Ǿ�������ؽ�����ijЩ����ɷ��ӵ������ǿ��еġ����о�����ƽ�ĸ���ӵ�Ӧ�ôӹ̶���ø�����һ����չ���˻�������Ϊ��ƽ�ĸ���ӵ�Ӧ�ÿ����˵�·��
����
�����������������˶����������������о��⣬��ʵ���Ҷ��ڽ�ĸ���ӹ̶���ø���о��ǽ�ĸ����Ӧ���о����ص㡣�������ҹ̶���ø�����ķ�չ����ƽ�ĸ���ӹ̶���ø��Ϊ���ҹ̶���ø�������³�Ա��������ص��ŵ��״�չ���ڴ����ǰ�� �̶���ø����װ“��Ȼ”��ɣ�������Ȼ�����ԣ���С��һ�����ظ�ʹ�ã���ɫ������“һ�߶�ø��ϵ”�Ѿ������������ȵȡ����������ŵ㼰�о���������ţ���ĸ���ӹ̶���ø����Ϊø�����ҹ̶����������ʣ�
����
������ �� �� �ף�
����
����[1] BENITA S. Microencapsulation: methods and industrialapplications [M]. CRC Press,2005.
����[2] PEYRATOUT C S,DAEHNE L. Tailor-made polyelectro-lyte microcapsules: from multilayers to smart containers[J]. Angewandte Chemie International Edition,2004,43�� 29�� : 3 762 -3 783.
����[3] MEIER W. Polymer nanocapsules [J]. Chemical SocietyReviews,2000,29�� 5�� : 295 - 303.
����[4] Ҧ������÷�ֺ͡� һ����������������ϵ�����������ʹ̶��������е�Ӧ�� [J]. Ĥ��ѧ�뼼����1999,19�� 1�� : 19 - 23.
����[5] HANEFELD U,GARDOSSI L,MAGNER E. Understand-ing enzyme immobilisation [J]. Chemical Society Re-views,2009,38�� 2�� : 453 - 468.
����[6] ��ʫһ�� ��Դ���ģ�������̼�ŷ����й���ҵ�Ŀɳ�����չ[J]. �����о���2009�� 4�� : 41 -55.
����[7] ���ʣ������������������ȡ� ��̼���õķ�չģʽ�о�[J]. �й��˿� ��Դ�뻷����2008,18�� 3�� : 14 -19.
����[8] NEIMAN A M. Ascospore formation in the yeast Saccharo-myces cerevisiae [J]. Microbiology and Molecular BiologyReviews,2005,69�� 4�� : 565 - 584.
����[9] NEIMAN A M. Sporulation in the budding yeast Saccharo-myces cerevisiae [J]. Genetics,2011,189 �� 3 �� : 737 -765.
����[10] SHI L,LI Z,TACHIKAWA H,et al. Use of yeast sporesfor microencapsulation of enzymes [J]. Applied and en-vironmental microbiology,2014,80 �� 15 �� : 4 502 -4 510.
����[11] ZHANG H,TACHIKAWA H,GAO X-D,et al. Appliedusage of yeast spores as chitosan beads [J]. Applied andenvironmental microbiology,2014,80 �� 16 �� : 5 098 -5 105.
����[12] LI Z,LI Y,DUAN S,et al. Bioconversion of d-glucoseto d-psicose with immobilized d-xylose isomerase andd-psicose 3-epimerase on Saccharomyces cerevisiae spores[J]. Journal of industrial microbiology & biotechnology,2015,42�� 8�� : 1 117 - 1 128.
����[13] CHEN S-C,DUAN K-J. Production of Galactooligosac-charides Using β-Galactosidase Immobilized on Chitosan-Coated Magnetic Nanoparticles with Tris �� hydroxymethyl��phosphine as an Optional Coupling Agent [J]. Interna-tional journal of molecular sciences,2015,16 �� 6 �� :12 499 - 12 512.
����[14] HONIGBERG S M. Ime2p and Cdc28p: Co-pilots drivingmeiotic development [J]. Journal of cellular biochemis-try,2004,92�� 5�� : 1 025 - 1 033.
����[15] LEE B,AMON A. Meiosis: how to create a specializedcell cycle [J]. Current opinion in cell biology,2001,13�� 6�� : 770 -777.
����[16] MITCHELL A P. Control of meiotic gene expression inSaccharomyces cerevisiae [J]. Microbiological reviews,1994,58�� 1�� : 56 - 70.
����[17] ROEDER G S. Sex and the single cell: meiosis in yeast[J]. Proceedings of the National Academy of Sciences,1995,92�� 23�� : 10 450 - 10 456.
����[18] STERN B M. FEARless in meiosis [J]. Molecular cell,2003,11�� 5�� : 1 123 - 1 125.
����[19] SUDA Y,RODRIGUEZ R K,COLUCCIO A E,et al. Ascreen for spore wall permeability mutants identifies a se-creted protease required for proper spore wall assembly[J]. PLoS One,2009,4�� 9�� : e7184.
����[20] TACHIKAWA H,BLOECHER A,TATCHELL K,et al.A Gip1p-Glc7p phosphatase complex regulates septin or-ganization and spore wall formation [J]. The Journal ofcell biology,2001,155�� 5�� : 797 - 808.
����[21] EGEL R. Selective spore survival during replica-plating offission yeast [J]. Archives of microbiology,1977,112�� 1�� : 109 -110.
����[22] KUPIEC M,BYERS B,ESPOSITO R E,et al. 11 Meio-sis and Sporulation in Saccharomyces cerevisiae [J]. ColdSpring Harbor Monograph Archive,1997,21: 889 -1 036.
����[23] BYERS B. Cytology of the yeast life cycle [J]. ColdSpring Harbor Monograph Archive,1981,11: 59 - 96.
����[24] STRATHERN J N,JONES E W,BROACH J R. Molecu-lar biology of the yeast saccharomyces,life cycle and in-heritance [M]. Cold Spring Harbor Laboratory,1981.
����[25] NAKANISHI H,MORISHITA M,SCHWARTZ C L,etal. Phospholipase D and the SNARE Sso1p are necessaryfor vesicle fusion during sporulation in yeast [J]. J CellSci,2006,119�� 7�� : 1 406 - 1 415.
����[26] MOENS P B. Fine structure of ascospore development inthe yeast Saccharomyces cerevisiae [J]. Canadian Journalof Microbiology,1971,17�� 4�� : 507 - 510.
����[27] LYNN R R,MAGEE P T. Development of the spore wallduring ascospore formation in Saccharomyces cerevisiae[J]. The Journal of cell biology,1970,44 �� 3�� : 688- 692.
�� [28] SMITS G J,VAN DEN ENDE H,KLIS F M. Differentialregulation of cell wall biogenesis during growth and devel-opment in yeast [J]. Microbiology,2001,147�� 4�� : 781- 794.
����[29] COOPER K F,MALLORY M J,EGELAND D B,et al.Ama1p is a meiosis-specific regulator of the anaphase pro-moting complex / cyclosome in yeast [J]. Proceedings ofthe National Academy of Sciences, 2000, 97 �� 26 �� :14 548 - 14 553.
����[30] OELSCHLAEGEL T,SCHWICKART M,MATOS J,etal. The yeast APC / C subunit Mnd2 prevents prematuresister chromatid separation triggered by the meiosis-specif-ic APC / C-Ama1 [J]. Cell,2005,120�� 6�� : 773 - 788.
����[31] PENKNER A M,PRINZ S,FERSCHA S,et al. Mnd2,an essential antagonist of the anaphase-promoting complexduring meiotic prophase [J]. Cell,2005,120�� 6�� : 789- 801.
����[32] COLUCCIO A,NEIMAN A M. Interspore bridges: a newfeature of the Saccharomyces cerevisiae spore wall [J].Microbiology,2004,150�� 10�� : 3 189 - 3 196.meiotic exit to cytokinesis during sporulation in Saccharo-myces cerevisiae [J]. Molecular biology of the cell,2009,20�� 1�� : 134 - 145.
����[33] DIAMOND A E,PARK J-S,INOUE I,et al. The ana-phase promoting complex targeting subunit Ama1 links
����[34] HIMURO Y,TAKAI M,ISHIHARA K. Poly �� vinylferro-cene-co-2-hydroxyethyl methacrylate�� mediator as immo-bilized enzyme membrane for the fabrication of ampero-metric glucose sensor [J]. Sensors and Actuators B:Chemical,2009,136�� 1�� : 122 - 127.
����[35] DEUTSCHBAUER A M,WILLIAMS R M,CHU A M,etal. Parallel phenotypic analysis of sporulation and post-germination growth in Saccharomyces cerevisiae [J]. Pro-ceedings of the National Academy of Sciences,2002,99�� 24�� : 15 530 -15 535.
����[36] COLUCCIO A,BOGENGRUBER E,CONRAD M N,etal. Morphogenetic pathway of spore wall assembly in Sac-charomyces cerevisiae [J]. Eukaryotic Cell,2004,3�� 6�� : 1 464 -1 475.
����[37] GOMEZ-ESQUER F,RODR�PGUEZ-PE�rA J M,D�PAZG,et al. CRR1,a gene encoding a putative transglycosi-dase,is required for proper spore wall assembly in Sac-charomyces cerevisiae [J]. Microbiology, 2004, 150�� 10�� : 3 269 -3 280.
����[38] SARKAR P,FLORCZYK M,MCDONOUGH K,et al.SSP2, a sporulation-specific gene necessary for outerspore wall assembly in the yeast Saccharomyces cerevisiae[J]. Molecular Genetics and Genomics,2002,267�� 3�� :348 - 358.
����[39] LI J,AGARWAL S,ROEDER G S. SSP2 and OSW1,two sporulation-specific genes involved in spore morpho-genesis in Saccharomyces cerevisiae [J]. Genetics,2007,175�� 1�� : 143 - 154.
����[40] CHU S,DERISI J,EISEN M,et al. The transcriptionalprogram of sporulation in budding yeast [J]. Science,1998,282�� 5389�� : 699 - 705.
����[41] CHRISTODOULIDOU A,BOURIOTIS V,THIREOS G.Two sporulation-specific chitin deacetylase- encodinggenes are required for the ascospore wall rigidity of Sac-charomyces cerevisiae [J]. Journal of Biological Chemis-try,1996,271�� 49�� : 31 420 - 31 425.
����[42] MISHRA C,SEMINO C E,MCCREATH K J,et al. Clo-ning and expression of two chitin deacetylase genes ofSaccharomyces cerevisiae [J]. Yeast,1997,13�� 4�� : 327- 336.[43] PAMMER M,BRIZA P,ELLINGER A,et al. DIT101�� CSD2,CAL1�� ,a cell cycle �\ regulated yeast gene re-quired for synthesis of chitin in cell walls and chitosan inspore walls [J]. Yeast,1992,8�� 12�� : 1 089 - 1099.
����[44] SANZ M,TRILLA O A,DURAN N,et al. Control ofchitin synthesis through Shc1p,a functional homologue ofChs4p specifically induced during sporulation [J]. Mo-lecular microbiology,2002,43�� 5�� : 1 183 - 1 195.
����[45] NEUWALD A F. Barth syndrome may be due to an acyl-transferase deficiency [J]. Current Biology, 1997,7�� 8�� : R462 - R6.
����[46] BRIZA P,ELLINGER A,WINKLER G,et al. Charac-terization of a DL-dityrosine-containing macromoleculefrom yeast ascospore walls [J]. Journal of BiologicalChemistry,1990,265�� 25�� : 15 118 - 15 123.
����[47] BRIZA P,KALCHHAUSER H,PITTENAUER E,et al.N,N' Bisformyl Dityrosine is an in vivo Precursor of theYeast Ascospore Wall [J]. European journal of biochem-istry,1996,239�� 1�� : 124 - 131.
����[48] BRIZA P,ECKERSTORFER M,BREITENBACH M.The sporulation-specific enzymes encoded by the DIT1and DIT2 genes catalyze a two-step reaction leading to asoluble LL-dityrosine-containing precursor of the yeastspore wall [J]. Proceedings of the National Academy ofSciences,1994,91�� 10�� : 4 524 - 4 528.
����[49] FELDER T,BOGENGRUBER E,TENREIRO S,et al.Dtr1p,a multidrug resistance transporter of the major fa-cilitator superfamily,plays an essential role in spore wallmaturation in Saccharomyces cerevisiae [J]. Eukaryoticcell,2002,1�� 5�� : 799 - 810.
����[50] ���㡣 ��ƽ�ĸ���ӹ̶���ø�� D-�����Ǻϳ� D-����ͪ�ǵ��о� [J]. 2015.
����[51] CHOW C K,PALECEK S P. Enzyme encapsulation inpermeabilized Saccharomyces cerevisiae cells [J]. Bio-technology progress,2004,20�� 2�� : 449 - 456.
����[52] ROMERO C,SANCHEZ S,MANJON S,et al. Optimi-zation of the pectinesterase / endo-D-polygalacturonase co-immobilization process [J]. Enzyme and microbial tech-nology,1989,11�� 12�� : 837 - 843.
����[53] MEN Y,ZHU Y,ZENG Y,et al. Co-expression of d-glucose isomerase and d-psicose 3-epimerase: develop-ment of an efficient one-step production of d-psicose [J].Enzyme and microbial technology,2014,64: 1 - 5.
����[54] MASARU S,MITSUTAKA Y. A new enzymatic serumcreatinine measurement based on an endogenous creatine-eliminating system [J]. Clinica chimica acta,1984,143�� 2�� : 147 -155.
����[55] FOSSATI P,PRENCIPE L,BERTI G. Enzymic creati-nine assay: a new colorimetric method based on hydrogenperoxide measurement [J]. Clinical chemistry,1983,29�� 8�� : 1 494 -1 496.
����[56] COLUCCIO A E,RODRIGUEZ R K,KERNAN M J,etal. The yeast spore wall enables spores to survive passagethrough the digestive tract of Drosophila [J]. PLoS One,2008,3�� 8�� : e2873.
��Ҫ��ʾ��ת�ر�վ��Ϣ��ע����Դ��985������������Ȩ�������������վ������
�Ķ���ʾ���������ж���Ϣ����ʵ�Լ��۵������վ�Ų�����
���ķ���

- �������Ŀ��ⱨ�淶�ļ���ʽҪ��
- ˶ʿ��ҵ���Ŀ��ⱨ�����Ҫ�Լ�
- �����������硷�еķ����а�����
- ��ҵ��ƿ��ⱨ�������ʽ������
- Ӣ�����Ŀ��ⱨ�淶�IJο�
- �Ŵ���ѧ���Ŀ��ⱨ�淶�IJο�
- ���̹���ѧλ���Ŀ��ⱨ����ôд
- дһƪ����ѧ˶ʿ���Ŀ��ⱨ��Ҫ
- Ӣ��˶ʿ���Ŀ��ⱨ��д������
- ������Դרҵ��ְ�о���д������
- ��ҵ�������Ʊ�ҵ�����Ŀ��ⱨ��
- ���̹���רҵ���ⱨ����ôд
- ������ְ�о������ⱨ��ע������
- һƪ3000�ֵIJ��������ְ�о���
- ��ҵ�������IJο�����д���ο���
���߿ͷ�

 ר��������ѯ
ר��������ѯ
